|
However, we’ve seen that IR spectroscopy can a great technique for identifying certain functional groups in an unknown molecule especially functional groups containing OH or CO. License: Creative Commons Attribution-ShareAlike 3.0 United States License. By itself, Infrared (IR) spectroscopy isn’t a great technique for solving the structure of an unknown molecule. Organic Chemistry 51B (UCI OpenCourseWare: University of California, Irvine). Required attribution: Vranken, David Van. Organic Chemistry (Chem 51B) is part of OpenChem.ĬHAPTER 13- Mass Spectrometry and Infrared SpectroscopyĠ4:42 - 13.1: The Mass Spectrometer-how does it work?Ġ9:26 - 13.1-13.3: Mass spectra are complex due to fragmentationġ1:55 - 13.4: The Exact Mass gives the Molecular Formulaġ5:14 - 10.2: Use the Molecular Formula to determine Degree of Unsaturationġ9:25 - 13.5: Electromagnetic Radiation and InfraredĢ2:45 - 13.6: Dumb Characteristics of an IR spectrumĢ6:56 - 13.6: Key Frequency Regions of the IR spectrumģ2:22 - 13.6: Practice: Regions of the IR spectrumģ4:58 - 13.6,7: Polar Bonds Lead to Intense Peaksģ9:35 - 13.7: Hydrogen bonding leads to lumpy peaksĤ2:06 - 13.6: Dumb Characteristics of an IR spectrum (revisited)Ĥ2:44 - 13.6,7: Check if C-H stretches are above or below 300 cm^(-1) This video is part of a 26-lecture undergraduate-level course titled "Organic Chemistry" taught at UC Irvine by Professor David Van Vranken. 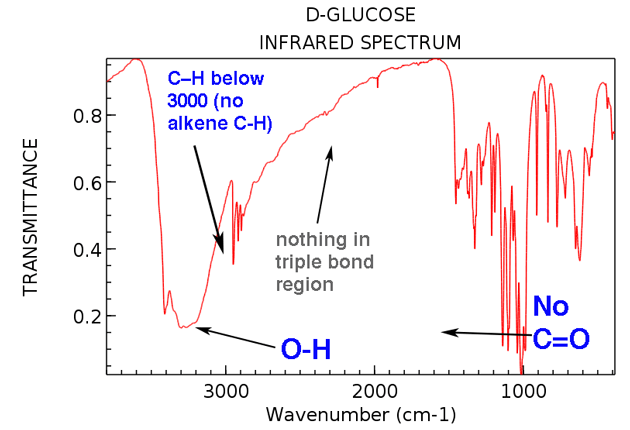  
Topics covered include: Fundamental concepts relating to carbon compounds with emphasis on structural theory and the nature of chemical bonding, stereochemistry, reaction mechanisms, and spectroscopic, physical, and chemical properties of the principal classes of carbon compounds. Organic Chemistry - Infared Spectroscopy and Chapter 14ĭescription: This is the second quarter of the organic chemistry series. UCI Chem 51B Organic Chemistry (Winter 2013)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
